Polarity Of The Bonds
Bonds molecules Polarity bond Chapter 5.6: properties of polar covalent bonds
Covalent Bonds: Predicting Bond Polarity and Ionic Character - Video
Polarity chemistry bonds Polarity bond polar powerpoint negative does positive cl which hcl ppt presentation Definition and examples of a polar bond
Polarity bond chemical bonding cl nacl hcl lewis theory ppt powerpoint presentation
Ionic bond covalent bonds character polarity bonding predicting chemical structure study matter ions lesson molecule plans atomic polyatomic resonance compoundPolar bonds covalent polarity bond vs molecular coordinate network molecule ppt powerpoint presentation negative positive Polar covalent bonds: electronegativity3.4: bond polarity.
Polarity of bonds and moleculesDefinition of bond polarity Polarity covalent polar electronegative differencesPolarity of bonds.

Polar molecule molecules lecture
Lecture 8.4b- polar moleculesPolarity laboratory molecular electron bonding Covalent bonds: predicting bond polarity and ionic characterPolarity bonding theories molecule atoms.
Polarity polar bonds molecules dipole molecule nonpolar priyamstudycentre chemical atomsPolar which diagram polarity bond molecule represents dipole moment electronegativity chemistry problems practice Covalent bonds nonpolar molecule molecularDifference between bond polarity and molecular polarity.

Bond polarity polar covalent bonds molecules ppt hcl atoms pair h2o ex powerpoint presentation electrons unequally pull shared
Polarity molecule molecules determiningChemistry 4.7 bond polarity Polar nonpolar δg calculateB polarity of bonds p18 difference in electronegativity — db-excel.com.
Ch. 8.4 – polar bonds and moleculesVsepr polarity bonds Difference between bond polarity and molecular polarityWhy are most organic molecules non-polar?.

Bond polarity between covalent molecular fluorine difference chemistry ionic carbon character polar cf vs bonding show atom form acid examples
Difference between polar and nonpolar examplesPolar molecules non organic most polarity bond why will bonds carbon hydrogen electronegative than they Polar bond covalent chemistry examples chemical bonds definition type types bonding example molecule molecules non nonpolar between science kids thoughtcoCovalent electronegativity bonds ionic character bonding.
Covalent bonds ionic bonding libretexts chapter polarity atoms electrons electron purely molecular structuresThe bond between which two atoms has the greatest degree of polarity in Solved:rank the bonds from most polar to least polar. a. c-o, c-f, c-nPolarity bonds.

Polar nonpolar bonds mcat chemistry dipole bond covalent ccl compound schoolbag info each chemical cl moments socratic will four bonding
Reading: covalent bondsFigure 3.10. ccl 4 is a nonpolar compound with four polar bonds Polarity bonds molecules bond presentation ppt powerpointVsepr, polarity, and bonds.
Atoms polarity molecule socraticElectronegativity polarity bonds p18 types Polarity bond molecular between difference summary definition🔴 polarity of bonds || chemistry for class 11||organic chemistry.

Which diagram best represents a polar molecule
Polar bonds most least rankBond bonding polar polarity electronegativity types covalent ionic chem nonpolar between chemistry electron character distribution chemical figure molecular libretexts differences .
.


Difference Between Bond Polarity and Molecular Polarity | Definition

Difference Between Bond Polarity and Molecular Polarity | Definition

Ch. 8.4 – Polar Bonds and Molecules
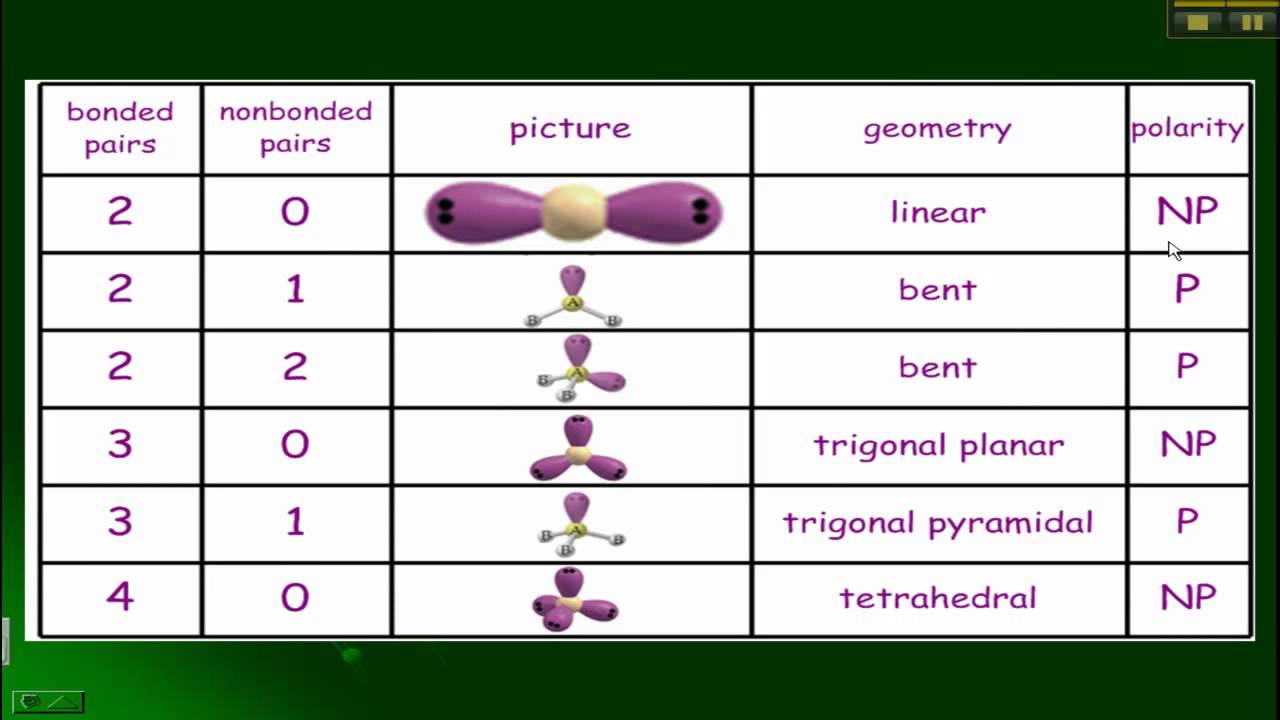
VSEPR, Polarity, and Bonds - YouTube

B Polarity Of Bonds P18 Difference In Electronegativity — db-excel.com

PPT - Polarity of Bonds & Molecules PowerPoint Presentation, free